Introduction
Radiotherapy (RT) is beneficial for select patients with localized diffuse large B cell lymphoma (DLBCL), in both the early stage and relapsed/refractory settings. Although molecular subtyping plays a key role in classification of DLBCL, genomic predictors of local response to RT have yet to be adequately explored. To our knowledge, there are no existing studies investigating the genomic landscape of DLBCL patients treated with RT. Therefore, we sought to identify genomic signatures of radiosensitivity in localized DLBCL patients.
Methods
We analyzed an institutional database of DLBCL patients treated with RT for localized measurable disease treated between 2007-2022. All patients underwent next-generation sequencing using an institutional 505-gene panel (MSK-Impact). Clinical and radiographic characteristics were obtained from chart and imaging review. All patients had measurable disease on CT scan at time of RT and included both Ann Arbor stage I/II patients referred for consolidative radiotherapy and those with isolated sites of relapsed/refractory disease. Patients did not receive any systemic therapy between RT start and time of response assessment. The public NCI LymphGen algorithm was used to further classify genomic subgroups. Using Fisher's exact test and logistic regression, we tested for univariable correlation between specific gene alterations and LymphGen subgroups with initial radiation response, defined by Lugano criteria and decrease in maximum SUV.
Results
We identified 46 patients including 15 (33%) tumors treated in the early-stage setting (13% definitively without prior chemotherapy, 87% as consolidation), while 31 (60%) were treated in the localized relapsed/refractory setting. The majority were GCB subtype (57%) while 28% were non-GCB and 15% had missing immunohistochemistry data. The median number of prior lines of systemic therapy was 1 (0-2) in early stage and 1 (1-7) in localized relapsed/refractory diseases. The median RT dose was 36 Gy (4-54), with 38 (83%) tumors receiving at least 30Gy.
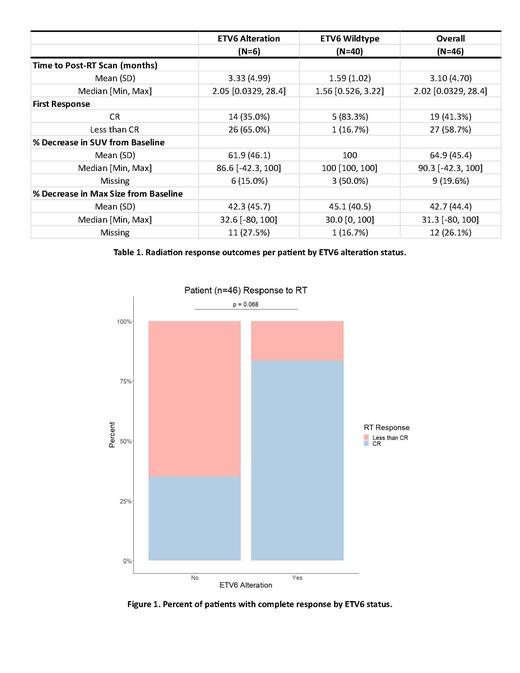
The median lesion SUV at the time of RT was 4.11 (1.1-46.0). Complete response (CR) was achieved in 41% of tumors at the time of first imaging assessment (median 3.1 months) with a median of 90.3% (42-100%) decrease in SUV. The most commonly altered individual genes were KMT2D (35%), MYD88 (28%), and TP53 (35%). LymphGen classified 22% of cases as EZB subtype, 13% as MCD, 2% as BN2, 2% as ST2, and the remaining 61% were unclassified. Mutations in ETV6 (13% of tumors) were associated with a higher CR rate in response to RT (83% mutated vs 35% wildtype, p=0.068). Lesions from ETV6 mutated cases had similar SUVs (p=0.74) and sizes (p=0.92) as wildtype cases. Alterations in genes from the LymphGen MCD subgroup were associated with greater reductions in SUV (ETV6 p<0.0001, PIM1 p<0.0001, MYD88 p<0.001, and TNFAIP3 p=0.02) The MCD subgroup trended towards increased odds of CR post-RT (log odds 1.9, p=0.08) and greater reduction of SUV (p=0.07) compared to the EZB subgroup.
Conclusions
To our knowledge this is the first study evaluating the role of LymphGen subgrouping on radiotherapy response, and the first study to examine genomic alterations in DLBCL patients treated with radiation. Genomic biomarkers of radiosensitivity are needed to identify radioresponsive tumors which may benefit from better integration of RT into existing salvage paradigms such as CAR T cells. Our data suggest that alterations in MCD LymphGen subgroup genes (ETV6, PIM1, MYD88, TNFAIP3) may be associated with better response to radiotherapy.
ETV6 is a strong hematopoietic transcription factor known to be altered in many human leukemias and myelodysplastic syndromes. Its role in DLBCL is not well defined, with preclinical studies suggesting a distinct role compared to leukemias. Due to the limited sample size, these findings are hypothesis generating and warrant further study with larger, prospective cohorts to validate the ability of genomic signatures to predict RT response.
Disclosures
Salles:Merck: Consultancy, Honoraria; Nurix: Consultancy; Orna: Consultancy; EPIZYME: Consultancy; Nordic Nanovector: Consultancy; Ipsen: Consultancy, Research Funding; Owkin: Current holder of stock options in a privately-held company; Loxo/Lilly: Consultancy; BeiGene: Consultancy; AbbVie: Consultancy, Honoraria; BMS/Celgene: Consultancy; ATB Therapeutics: Consultancy; Janssen: Consultancy, Research Funding; Debiopharm: Consultancy; Genentech, Inc./F. Hoffmann-La Roche Ltd: Consultancy, Research Funding; Molecular Partners: Consultancy; Kite/Gilead: Consultancy; Genmab: Consultancy; Novartis: Consultancy; Incyte: Consultancy. Joffe:Abbvie: Honoraria; Beigene: Honoraria. Imber:GT Medical Technologies: Honoraria. Yahalom:Convergent R.N.R Ltd.: Other: Provision of Services (uncompensated).

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal